AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Periodic table and reactivity3/20/2024   /chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png) 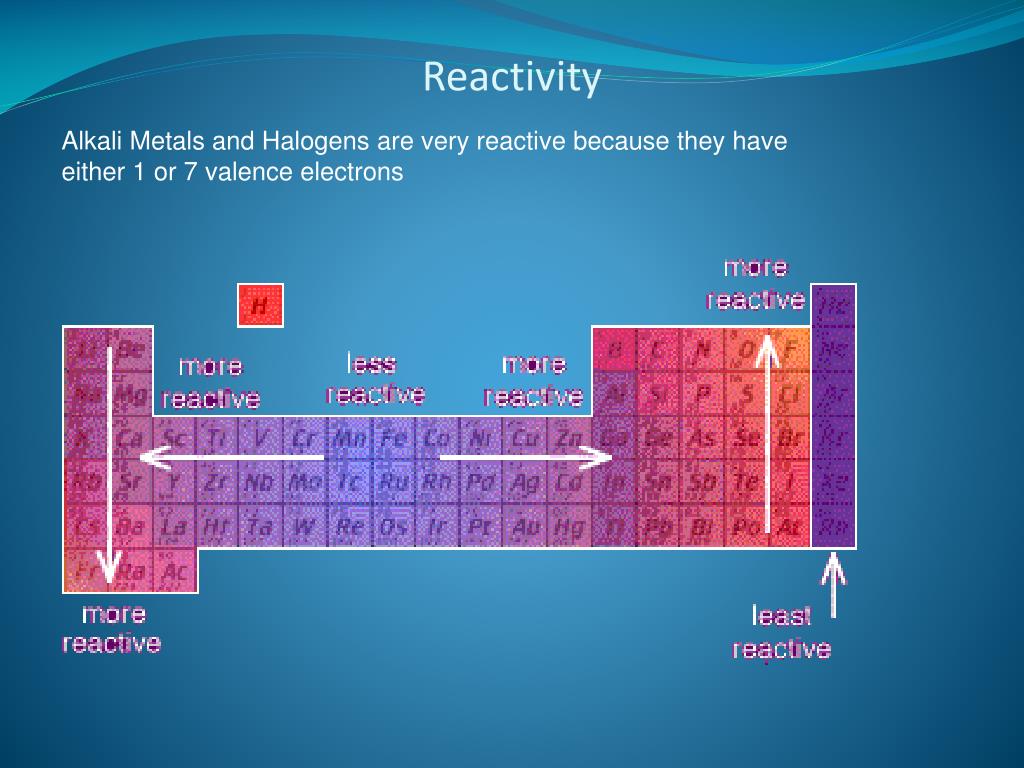
reaction with oxygen is slow at room temperature, but they can ignite around 150-200 ☌. The elements on the far left of the periodic table react with oxygen to generate basic. The Lanthanides have the following reactions: oxidize rapidly in moist air. Oxides are formed when elements interact with oxygen. Gethin: So, the further down the table you go the more reactive they become. Chemical Reactivity (Also Known As Reactivity) It is possible to gain a comprehensive understanding of an element’s chemical reactivity by observing the reaction of the element with oxygen and halogens. So, the reason we didn't see the reactions of those last three metals is that they are so reactive the explosion would be far too big. Miss Armit: No, it's the second rarest element on Earth. That's not an element that you hear a lot of. It consists of the elements oxygen, sulfur, selenium, tellurium and polonium. The oxygen family, also called the chalcogens, consists of the elements found in Group 16 of the periodic table and is considered among the main group elements. The general reaction for alkali metal hydrides and water is given below: (4) M H ( s) + H 2 O ( l) M ( a q) + + O H ( a q) + H 2 ( g) This reaction can be. In this case, however, hydrogen gas is produced with the metal hydroxide. We saw there lithium, then sodium and then potassium, but you didn't see rubidium, caesium and francium. Group 16: General Properties and Reactions. Similarly to the Group 1 oxides, the hydrides of the Group 1 elements react with water to form a basic solution. As you can see the metal's get increasingly more reactive as you go down this group. Gethin: It's fair to say that you love the Group 1 metals, don't you. Well, sodium is below lithium in group one and the reactivity of the alkali metals increases as you go down the group, so potassium is even more reactive. So let's try it with another Group 1 metal - sodium.Īs you can see it reacts far more vigorously then lithium. And can you see the little purple trail there? That universal indicator is showing us that the water now contains an alkali in this case - lithium hydroxide. It's transferring its energy to its surroundings by heating. Scientists are working on creating and verifying. Elements arent discovered or created in order of atomic number. The present periodic table has room for 118 elements. The alkali metals, alkaline earths, basic metals, transition metals, lanthanides, and actinides all are groups of metals. Well, you can see it's reacting with the water straight away - it's steadily fizzing away there. Most of the elements on the periodic table are metals. And if I put some universal indicator in, like this, you'll see that it's green and that means it's neutral.īut let's see what happens when we put the lithium in. And do you see how quickly it tarnishes, how quickly it's changing colour? Here we've got some water. 
Here I've got some lithium and I'm just going to cut a little bit off. When a Group 1 element reacts with water it produces a metal hydroxides solution and hydrogen gas. They're soft and shiny when freshly cut, but they quickly tarnish as the metal reacts with oxygen in the air. Let's take a look at some Group 1 metals then - also known as alkaline metals.įran: Group 1 elements are called the alkali metals. For example, a quick glance at Figure AT5.2.Gethin: And today we're going to be looking at the periodic table. predict which element forms the most stable compounds in the +2 oxidation state. That way, we can get a better look at the relationship. Based on the positions of the group 14 elements C, Si, Ge, Sn, and Pb in the periodic table and the general trends outlined in this section, classify these elements as metals, semimetals, or nonmetals. Often it is useful to plot data on a graph. Is as follows: To remember how the reactivity of the alkali metals and halogens increases or decreases, put a pin in the. As you go up group 7 (the halogens), again the elements get more reactive. In chemistry, reactivity is the impulse for which a chemical substance undergoes a chemical reaction, either by itself or with other materials, with an overall release of energy. As a result, fluorine is usually thought of as the most electronegative element. A useful mnemonic picture to help you recall that: As you go down group 1 (the alkali metals) in the periodic table, the elements get more reactive. However, on many scales, fluorine would be the most electronegative atom here. As a result, noble gases are also given electronegativity values on this scale. The Allen scale just depends on the ability of an atom to interact with light, which is something even noble gases can do. Some electronegativity scales do not have values for the noble gases, because they are based on experimental measurements of compounds, and noble gases do not commonly form compounds with other elements. \): The Allen electronegativity values of the second-row elements. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
